SINGLE-MOLECULE SPECTROSCOPY GROUP
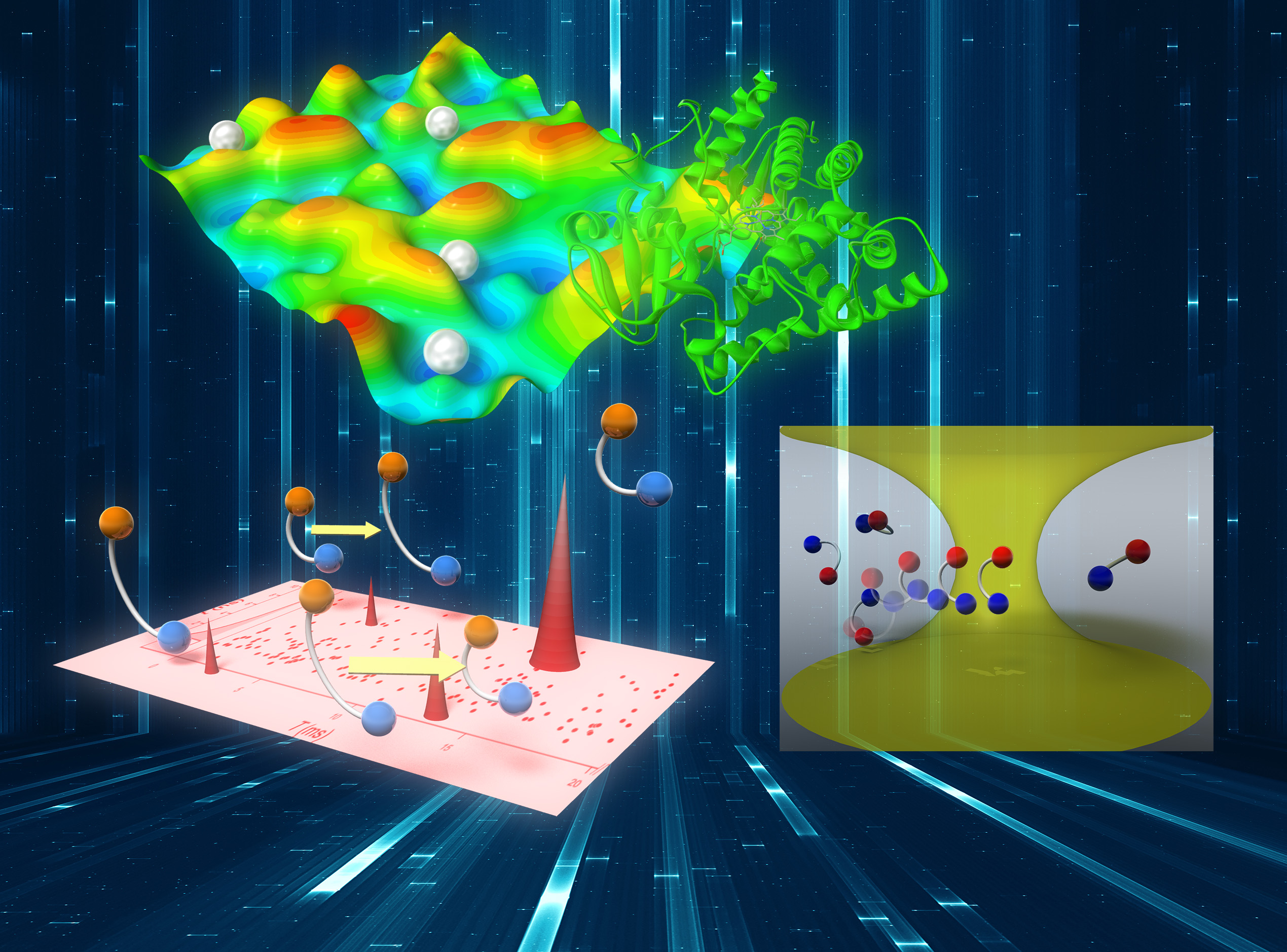
Development of novel single-molecule methods and application to elucidate complex behavior of biomolecular systems
 Kunihiko Ishii
Kunihiko Ishii
Research outline
Biopolymers (e.g., proteins and nucleic acids) and biomolecular assemblies (e.g., lipid membranes) are important classes of molecular systems which constitute life. Since they consist of a large number of atoms and have a high structural degree of freedom, such complex biomolecular systems flexibly change their structures under room temperature. Behind this flexibility, there exists a structural hierarchy, and correspondingly, their dynamics reveals a broad range of characteristic time scales. This hierarchy leads to structural heterogeneity and complex dynamics of the biomolecular systems, which, in turn, plays crucial roles in the biological functions. To examine the structural heterogeneity and spontaneous dynamics of complex biomolecular systems, an attractive and powerful way is to measure single molecules one by one, instead of observing the average response of many molecules. However, because in general, the spectroscopic signal emitted from a single molecule is extremely faint, it is not a simple task to know much about the molecular property through the observed single-molecule data. We are developing novel experimental and analytical tools having high information content based on time-resolved single-molecule fluorescence spectroscopy and photon correlation analysis. We aim at elucidating the complex behavior of biomolecular systems through making use of these original methods.
Projects
Development of two-dimensional fluorescence lifetime correlation spectroscopy
Study of biomacromolecular dynamics with a microsecond time resolution using two-dimensional fluorescence lifetime correlation spectroscopy
Members

Kunihiko Ishii (group leader)
Tsukasa Tokita
Sandeep Yadav
Selected publications
- W. Heo; K. Hasegawa; K. Okamoto; Y. Sako; K. Ishii; T. Tahara Scanning two-dimensional fluorescence lifetime correlation spectroscopy: Conformational dynamics of DNA Holliday-junction from microsecond to sub-second J. Phys. Chem. Lett. 13, 1249-1257 (2022).
- B. Sarkar; K. Ishii; T. Tahara Microsecond folding of preQ1 riboswitch and its biological significance revealed by two-dimensional fluorescence lifetime correlation spectroscopy J. Am. Chem. Soc. 143, 7968-7978 (2021).
- C. Cheng; K. Ishii; T. Tahara Microsecond equilibrium dynamics of hairpin-forming oligonucleotides quantified by two-color two-dimensional fluorescence lifetime correlation spectroscopy J. Phys. Chem. B 124, 10673-10681 (2020).
- B. Sarkar; K. Ishii; T. Tahara Microsecond conformational dynamics of biopolymer revealed by dynamic-quenching two-dimensional fluorescence lifetime correlation spectroscopy with single dye-labeling J. Phys. Chem. Lett. 10, 5536-5541 (2019).
- T. Otosu; K. Ishii; T. Tahara Microsecond protein dynamics observed at the single molecule level Nat. Commun. 6, 7685 (2015).
- K. Ishii; T. Tahara Two-dimensional fluorescence lifetime correlation spectroscopy. 2. Application J. Phys. Chem. B 117, 11423-11432 (2013).
- K. Ishii; T. Tahara Two-dimensional fluorescence lifetime correlation spectroscopy. 1. Principle J. Phys. Chem. B 117, 11414-11422 (2013).
